
Extracting Tin from Cassiterite — Smelting the Bronze Alloying Metal
Leiðbeiningar
Identify and Crush Cassiterite
Identify and Crush Cassiterite
Cassiterite (SnO2) is a heavy, dark brown to black mineral with an adamantine (diamond-like) lustre. It is typically found in alluvial stream deposits (stream tin) or in hard rock veins associated with granite. Alluvial cassiterite can be concentrated by panning, similar to gold — it is dense (density 7.0 g/cm3) and sinks readily in water. Crush the ore to small pieces (5-10 mm) using a hammerstone. For stream tin, the cassiterite is already in small, rounded pebbles that may only need light crushing. Cassiterite contains approximately 78 percent tin by weight, making it a remarkably rich ore.
Efni fyrir þetta skref:
Nauðsynleg verkfæri:
 Bellows
Bellows Hammerstone
Hammerstone Forge Tongs
Forge TongsSet Up the Smelting Furnace
Set Up the Smelting Furnace
Tin smelting requires lower temperatures than copper smelting — tin melts at only 232 degrees C, and cassiterite reduces at approximately 800-900 degrees C. A small clay furnace or even a deep crucible nested in a bed of charcoal with forced air is sufficient. Line the furnace or crucible with a layer of charcoal at the bottom. Tin smelting can also be done in a simple bowl furnace — a shallow clay-lined pit with a tuyere for bellows air. The lower temperature requirement means that tin smelting was likely discovered before copper smelting, possibly when cassiterite-bearing rocks were used in campfire rings.
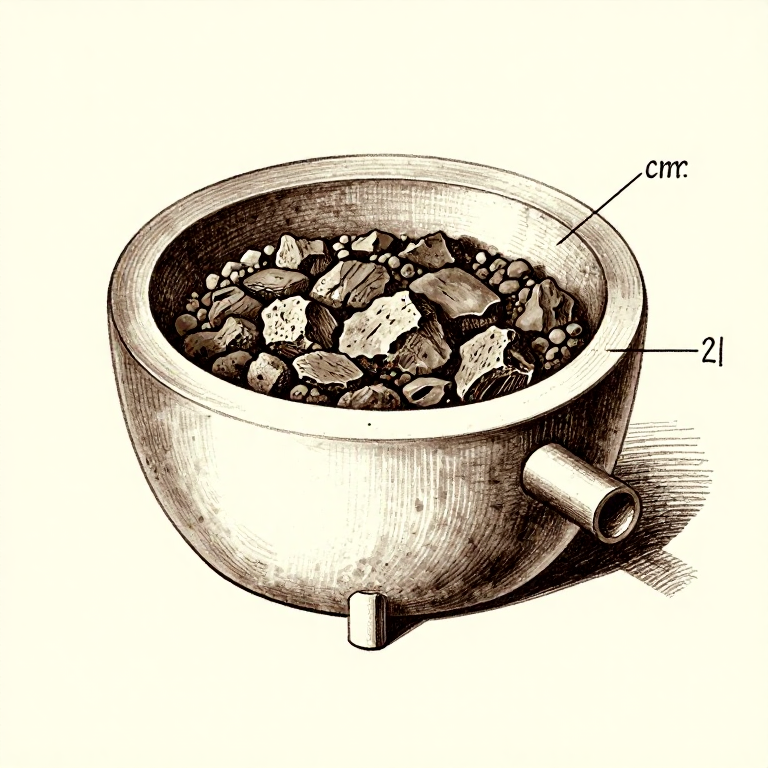
Charge and Smelt
Charge and Smelt
Layer the crushed cassiterite with charcoal in the furnace — approximately one part ore to three parts charcoal by volume. Light the charcoal and begin pumping the bellows to raise the temperature above 800 degrees C. The reduction reaction proceeds as: SnO2 + 2CO -> Sn + 2CO2. The carbon monoxide from the burning charcoal strips the oxygen from the tin oxide, leaving metallic tin. Because tin has such a low melting point (232 degrees C), the reduced tin immediately melts and collects at the bottom of the furnace as a liquid pool. Continue the smelt for 1-2 hours to ensure complete reduction of the ore.
Collect the Tin
Collect the Tin
Allow the furnace to cool slightly, then carefully tilt or break it to pour out the molten tin. Alternatively, if using a crucible, lift it with tongs and pour the tin into a stone or clay mould. Liquid tin is silvery-white and very fluid. As it solidifies, pure tin makes a characteristic crackling sound known as the 'tin cry' — this audible cracking is caused by the mechanical twinning of tin's crystal structure during deformation and is a simple field test for tin purity. Separate the tin from any adhering slag (which floats on top due to lower density) by re-melting and skimming.
Alloy with Copper for Bronze
Alloy with Copper for Bronze
The primary use of tin in antiquity was alloying with copper to produce bronze. The standard bronze alloy is approximately 90 percent copper and 10 percent tin by weight. Melt copper in a crucible first (it has the higher melting point at 1085 degrees C), then add the tin to the molten copper. Stir the mixture with a green stick to ensure thorough mixing. The resulting bronze is significantly harder than pure copper, has a lower melting point (approximately 950 degrees C, making it easier to cast), and flows more readily into moulds. The importance of tin to Bronze Age civilisations cannot be overstated — tin sources in Cornwall, Afghanistan, and Southeast Asia drove maritime and overland trade routes spanning the entire ancient world.
Efni
3- 1-3 kg crushed pieceStaðgengill
- 1 pieceStaðgengill
Nauðsynleg verkfæri
3- Staðgengill
- Staðgengill
- Staðgengill
CC0 opinbert ríki
Þessi teikning er gefin út undir CC0. Þér er frjálst að afrita, breyta, dreifa og nota þetta verk í hvaða tilgangi sem er, án þess að biðja um leyfi.
Studdu smiðinn með því að kaupa vörur í gegnum teikningu hans þar sem hann fær þóknun smiða sem seljendur ákvarða, eða búðu til nýja endurskoðun á þessari teikningu og tengdu hana sem tengingu í þinni eigin teikningu til að deila tekjum.